 But even when treatment is successful and hair regrows, hair loss may return once treatment stops. If treatment works, it can take up to six months to regrow hair in affected areas. Learn more: All about off-label prescription drug use » So, your doctor can prescribe a drug however they think is best for your care. This is because the FDA regulates the testing and approval of drugs, but not how doctors use drugs to treat their patients. However, a doctor can still use the drug for that purpose. 
Off-label drug use means that a drug that’s been approved by the FDA for one purpose is used for a different purpose that has not been approved. Food and Drug Administration (FDA) for treating rheumatoid arthritis. 
However, this is considered off-label use of tofacitinib, which is approved by the U.S. Tofacitinib (Xeljanz) appears highly effective for AU. A high level of immune reactivity is present in patients with alopecia totalis and alopecia universalis than in normal controls 9. Your doctor may also suggest ultraviolet light therapy to promote blood circulation and activate hair follicles. Some patients do respond well to currently available treatments, but the response rate in those with severe alopecia areata types (alopecia totalis, alopecia universalis or a combination) remains low. Both therapies help activate hair follicles and promote hair growth. This is believed to redirect immune system response away from hair follicles. 'Alopecia' is a Latin term that means baldness, and 'areata' refers to the patchy nature of the hair loss that is typically seen with this condition. Topical diphencyprone produces an allergic reaction to stimulate an immune system response. Description Collapse Section Alopecia areata is a common disorder that causes hair loss. Topical immunotherapies stimulate the immune system. You may also be given topical treatments. This condition is classified as an autoimmune disease, so your doctor may recommend corticosteroids to suppress your immune system. Alopecia universalis congenita (ALUNC) is a severe autosomal recessive form of alopecia characterized by a complete absence of hair development affecting. Because AU is a severe type of alopecia, success rates vary. In some cases, treatment can restore hair to affected areas. FDA requires warnings about increased risk of serious heart-related events, cancer, blood clots, and death for JAK inhibitors that treat certain chronic inflammatory conditions.The goal of treatment is to slow or stop hair loss. The etiology is unknown, but evidence is. Breakthrough therapy.įood and Drug Administration. Alopecia areata is a common form of nonscarring, usually patchy hair loss affecting up to 2 of the population. 
Olumiant label.įood and Drug Administration. Montagna Symposium 2017 - Janus Kinase inhibitors for treatment of alopecia areata. Wang EHC, Sallee BN, Tejeda CI, Christiano AM. Seasonal patterns in alopecia areata, totalis, and universalis. Race and Alopecia Areata amongst US Women. Role of family history in patchy alopecia areata. Wang S, Ratnaparkhi R, Piliang M, Bergfeld WF. Pratt CH, King LE Jr, Messenger AG, Christiano AM, Sundberg JP. Different trichoscopic features of tinea capitis and alopecia areata in pediatric patients. Alopecia areata incognita and diffuse alopecia areata: clinical, trichoscopic, histopathological, and therapeutic features of a 5-year study. 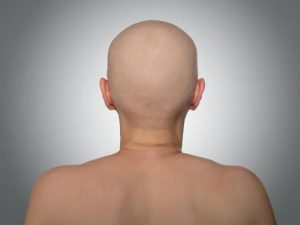
Alessandrini A, Starace M, Bruni F et al. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
